Manual handling refers to any activity that requires us to use force to lift, push, …
Read More »Boiler and Mechanical Power
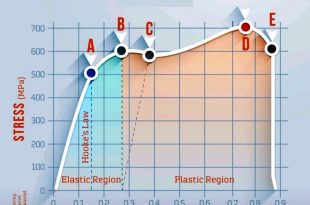
Hooke’s law Stress Strain Diagram Explanation
Introduction Hooke’s law describes the relationship between stress and strain in a material, particularly in …
Read More »Boiler Mountings and Accessories
Boiler Mountings and Accessories are integral to the functioning of steam boilers. These components play …
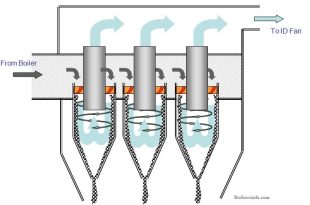
Read More »Boiler Grit Arrestor Working Principle
Boiler Grit Arrestor Working Principle: Ensuring Efficient Boiler Operation In the world of industrial and …
Read More »Classification and Types of Pumps
¨Pumps are divided into two fundamental types based on the manner in which they transmit …
Read More »Turbine Supervisory Instrumentation Enhancing Efficiency and Reliability
Gas or Steam Turbines are essential equipment in various industries, including power generation, oil and …
Read More »Understanding Air Pollution Emissions and Flue Gas Emission Standards in Boiler Combustion
Introduction: Boilers are widely used in industries and households for various purposes, including heating, power …
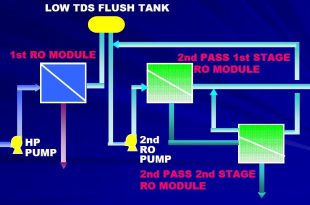
Read More »RO Plant Reverse Osmosis System
Introduction When it comes to obtaining clean and purified water, the reverse osmosis (RO) system …
Read More »Types of Measuring Tools
Types of Measuring Tools Measuring tools are essential for precise and accurate measurements in various …
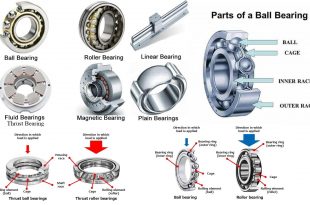
Read More »Types of Bearings and Their Applications
Bearings are mechanical components that reduce friction between moving parts and support loads. They come …
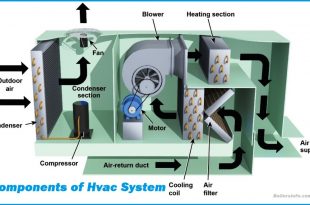
Read More »The Main Components of Hvac System
HVAC (Heating, Ventilation, and Air Conditioning) is an essential system for creating a comfortable indoor …
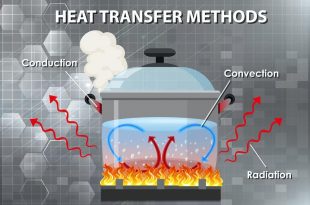
Read More »Methods of Heat Transfer
Heat transfer is the movement of thermal energy from one object or system to another. …
Read More »NDT Nondestructive Testing Methods
Nondestructive Testing (NDT) is a group of inspection techniques used to evaluate the properties of …
Read More »Burner Management System BMS
A burner management system (BMS) is a control system that is used to monitor and …
Read More »
Boiler and Mechanical Power Website
Welcome to Boilersinfo (Boiler and mechanical power) website is the Biggest Boiler Digital Library a collection of 3000+ FREE books and articles. We provide free training courses and study material related to boilers and mechanical power plants Oil and Gas Petroleum Books. visit the books section you will find hundreds of pdf doc ppt format documents for direct download.
This section is consists of 3000+ free documents books related to Mechanical Engineering, Power Plants, Boiler Engineering, and steam generators Operation, Maintenance theory, and practicals. section dived into many categories. The workshop, HVAC, Steam Turbines Welding, Thermodynamics, HSE fire Safety, and Electrical Engineering, etc books also included. Basic to advance Power plant books and the MCQ also added in this section.
In this Boiler and mechanical power web videos section you will find free paid training videos and courses about Boilers, Power Plants, Pumps, Valves, Process plant utilities, HSE industrial Safety Workshop, Bearings, and Electrical Training Videos.
This section is consists of hundreds of articles Regarding Boilers and Power plant technology, High-pressure Boiler Commissioning operation, and maintenance. Articles on Boiler safety and industrial safety HSE Process and power plant utilities and various Mechanical topics and are also included in separate sections.
Boiler & Mechanical Engineering Group
Join us and Register in our Boiler Engineering Community Forums. It is a community of Mechanical Engineers, Boiler Engineers operators, and Technicians professionals across the world. We will discuss and share knowledge about Boilers Operation, Maintenance, and Power plants as well as Mechanical engineering Welding, HVAC, HSE Safety, thermodynamics Boiler Examinations training, and study materials books slides.
-
Methods of Heat Transfer
Heat transfer is the movement of thermal energy from one object or system to another. …
Read More » -
Fire Tube Boiler
-
Boiler Design Basic Steps
-
Types and Classifications of Industrial Boilers
-
Boiler Hydrostatic testing procedure
-
Water Tube Boiler Parts and Functions
-
Boiler Mountings and Accessories
-
Pressure Safety Valve theory and PSV Valve Mechanism
-
Steam Boiler Alkali Boil out Procedure
-
Steam Blowing Procedure
-
RO Plant Reverse Osmosis System
-
Stoker Fired Boiler types of stokers
-
Role of Chemistry in a Power Plant
-
Combined Cycle Power Plant CCPP
-
Boiler Feed Water Pump Working Principle and Operation
 Boilersinfo Boiler and Mechanical Power Digital Library
Boilersinfo Boiler and Mechanical Power Digital Library